▍March 25, 2026 · Guangzhou
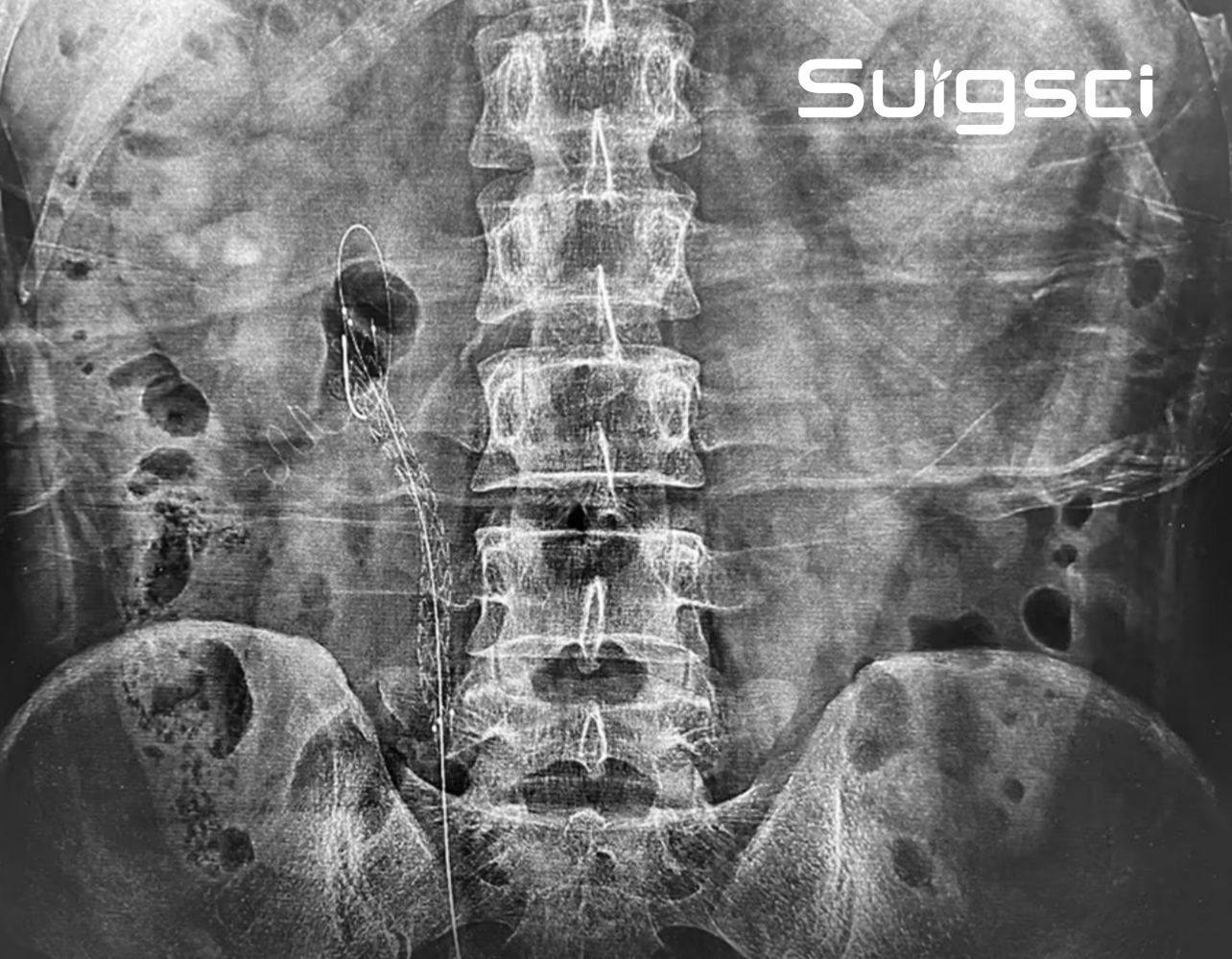
On March 25, 2026, Professor Xu Guibin from the Fifth Affiliated Hospital of Guangzhou Medical University successfully performed the first clinical implantation of the MimoPath™ ureteral covered stent (Mimo™ stent) in China.
Designed with a detachable, polymer-covered metal structure and controlled tear-line technology, the stent addresses key clinical challenges in the treatment of complex ureteral strictures and marks an important step forward in ureteral device development.
▍Core Technologies
The Mimo™ stent is fully independently developed by Surgsci, from materials and processes to proprietary equipment. As a leading innovative metal‑covered ureteral stent with advanced integrated technologies, it delivers the following key advantages:
miCot™ Coating Technology: The coating provides excellent adhesion to the stent substrate and a hydrophobic surface, which together reduce biofilm attachment and improve long‑term safety.
unLoad™ Detachment Technology: The detachment force is precisely engineered to provide a reliable balance between secure retention and easy, atraumatic removal.
Extended Size Range: Comprehensive sizing with a maximum working length exceeding 200 mm, among the industry’s longest. Enables single‑stent coverage of longer strictures for complex anatomy.
Low‑Friction Surface Treatment: Ensures smoother insertion and reduces deposition on the stent surface, extending its effective indwelling duration.
Segment‑Specific Reinforcement Design: Provides targeted radial strength at the stricture site while maintaining a wide lumen for unrestricted urine drainage, ensuring better conformity to complex ureteral anatomy.
▍Clinical Significance
The significance of this first clinical implantation extends far beyond the completion of a single procedure.
Complex ureteral strictures present significant clinical challenges, including long lesion segments, complex anatomy, short indwelling time of conventional stents, limited drainage performance, and material deposition that affects clinical outcomes.
The Mimo™ stent is engineered to address these unmet needs with long-term indwelling capability, safe and controlled removal, smooth delivery, and targeted support at the stricture site.
Treatment options for complex ureteral strictures have long been limited by conventional stent performance. The successful implantation of the Mimo™ stent represents a major breakthrough, offering urologists an innovative, high-performance solution for complex cases and marking a key milestone in ureteral device innovation.
▍About Surgsci
As the full‑stack developer of the MimoPath™ stent, Surgsci continues to innovate in minimally invasive urology devices. This clinical milestone reflects our engineering strength and our commitment to better patient care.
Built on our in-house R&D and standardized manufacturing, Surgsci delivers advanced urological solutions to clinicians and partners across the globe.